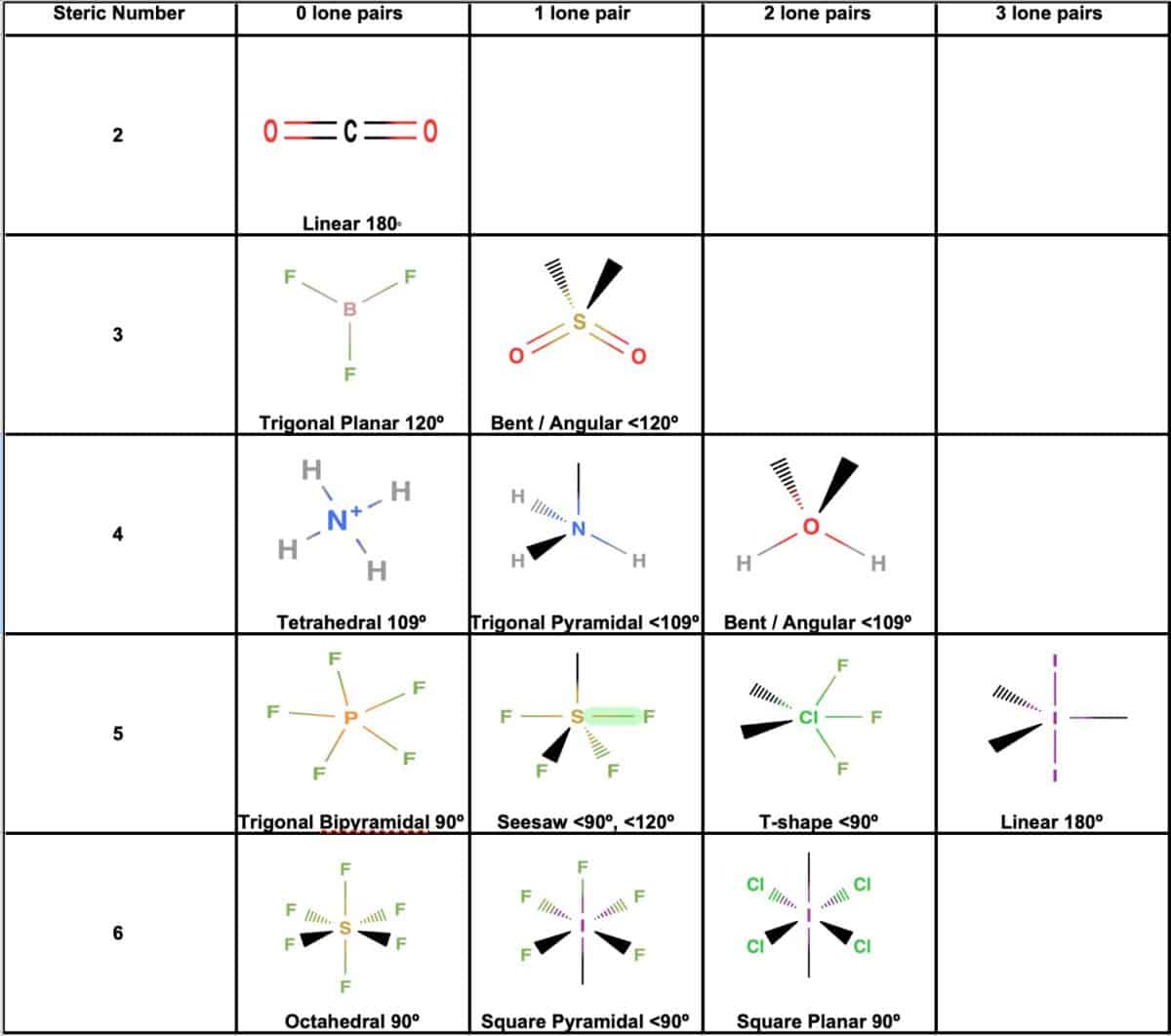
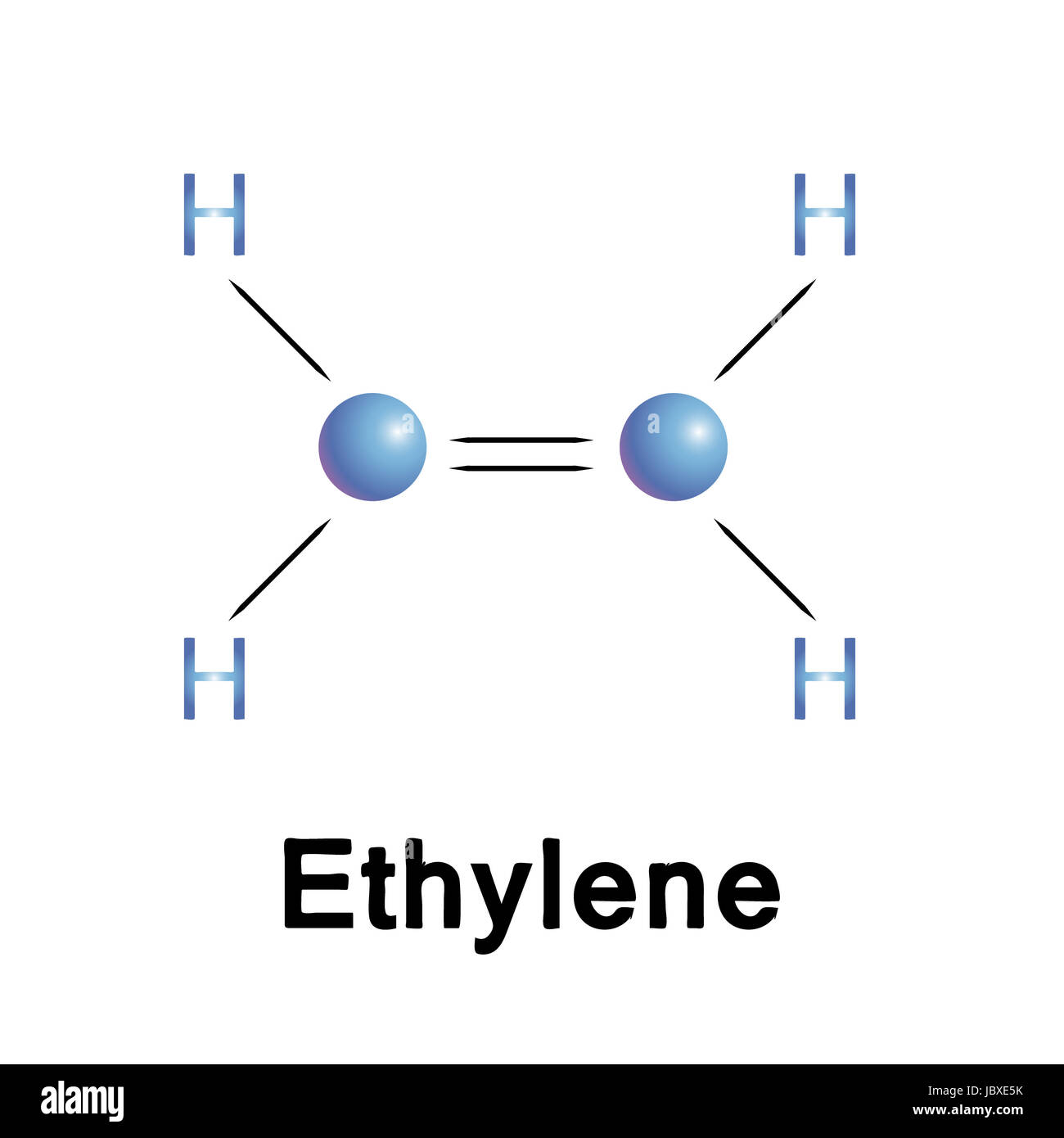
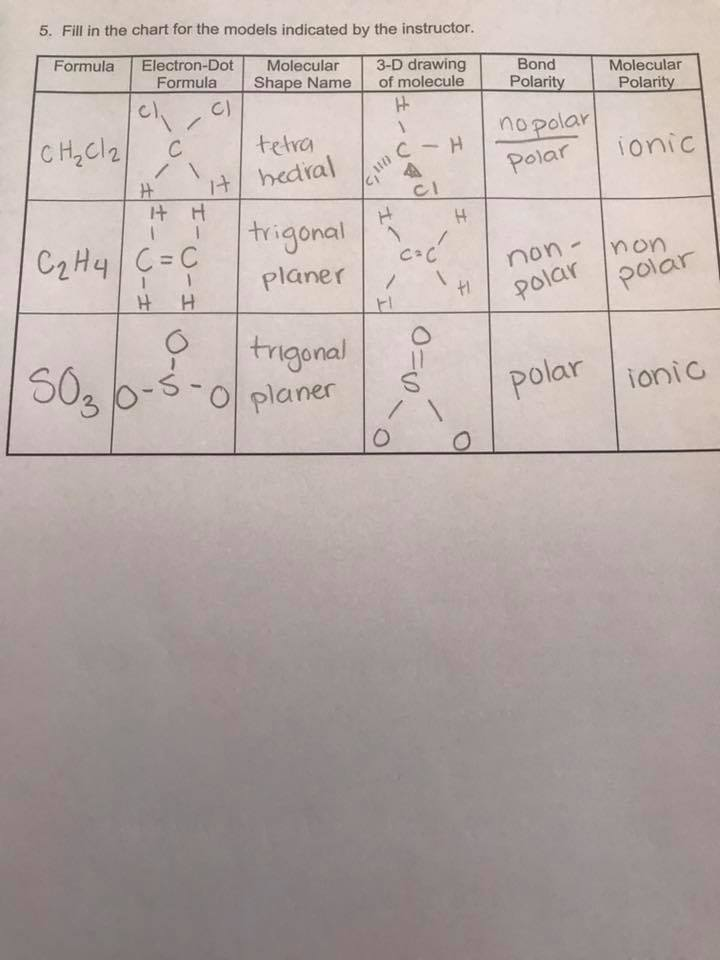
Each carbon atom is sp 2 hybridized, meaning they have three regions of electron density (one double bond and two single bonds). This results in a … · in this article, you will learn the easiest method to draw the lewis dot structure of ethene (c2h4). Learn how to draw the structure and understand its … All you need to know about c2h4: · explore the lewis structure of ethylene (c2h4) and uncover its bonding pattern, molecular geometry, and properties. A quick explanation of the molecular geometry of c2h4 including a description of the c2h4 bond angles. looking at the c2h4 lewis structure we can see that the. Additionally, you will find other interesting facts about the molecular geometry … - c2h4 lewis structure - shape (molecular geometry) - polarity Learn how to draw the lewis structure for c2h4 (ethene) and understand the bonding and molecular geometry of this important organic compound. · master the c2h4 lewis structure with our step-by-step guide and detailed diagram. · a quick explanation of the molecular geometry of c2h4 including a description of the c2h4 bond angles. looking at the c2h4 lewis structure we can see that the. The geometry of c 2 h 4 is planar. Ethylene is an essential industrial ingredient that is used in several applications. Read this article on c2h4 to find out its lewis structure, hybridization, molecular … Learn how to draw the ethylene molecule, understand its electron geometry, and explore its … · in the c2h4 lewis structure, there is a double bond between the two carbon atoms, and each carbon is attached with one hydrogen atom, and none of the atoms
The C2H4 Molecular Geometry Guide: Master It In 5 Minutes (And Save Your Job!)
Each carbon atom is sp 2 hybridized, meaning they have three regions of electron density (one double bond and two single bonds). This results in...