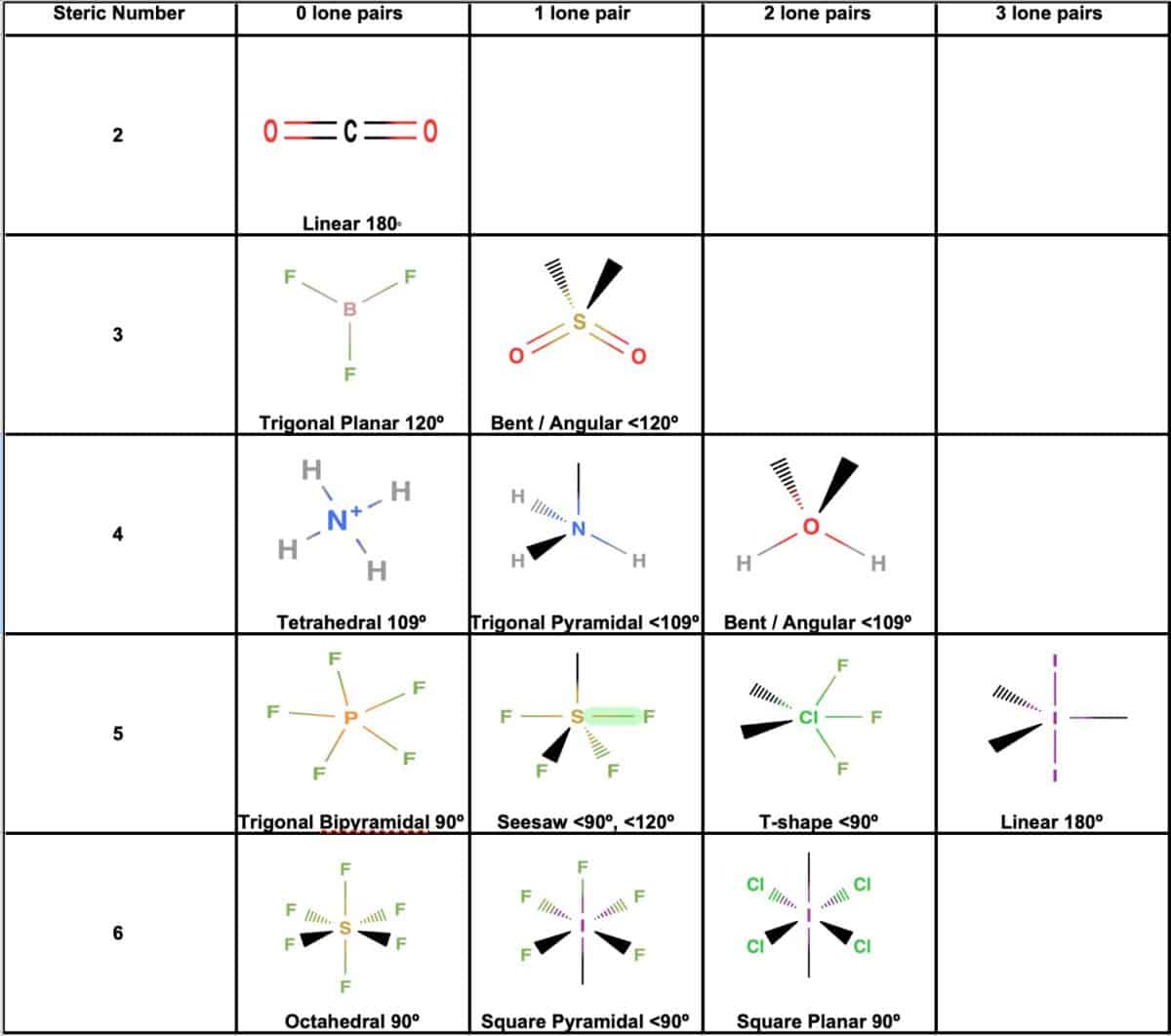
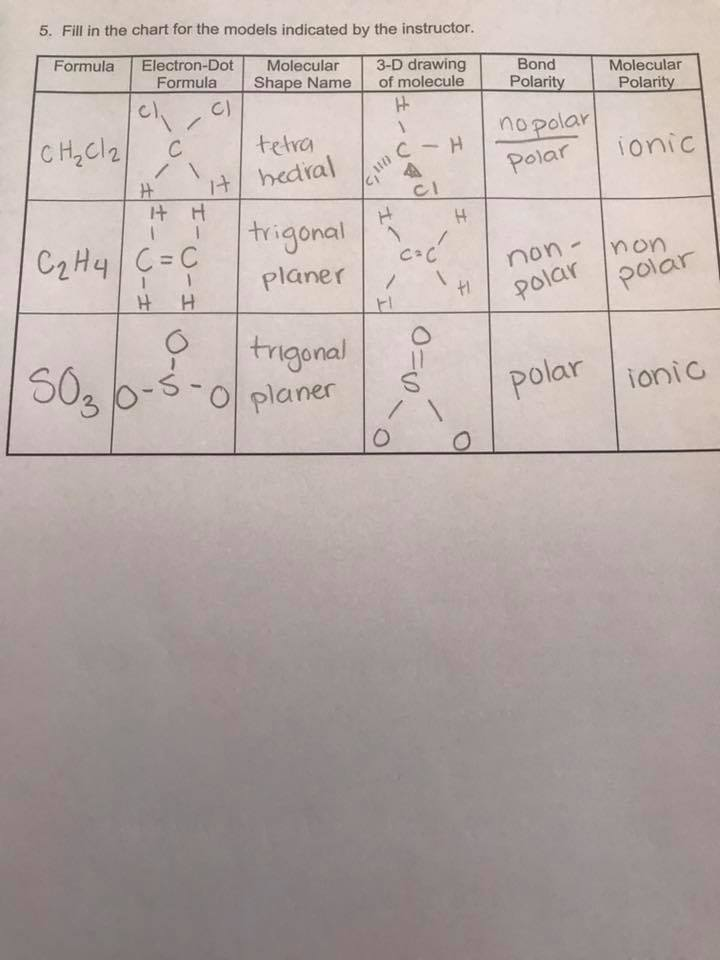
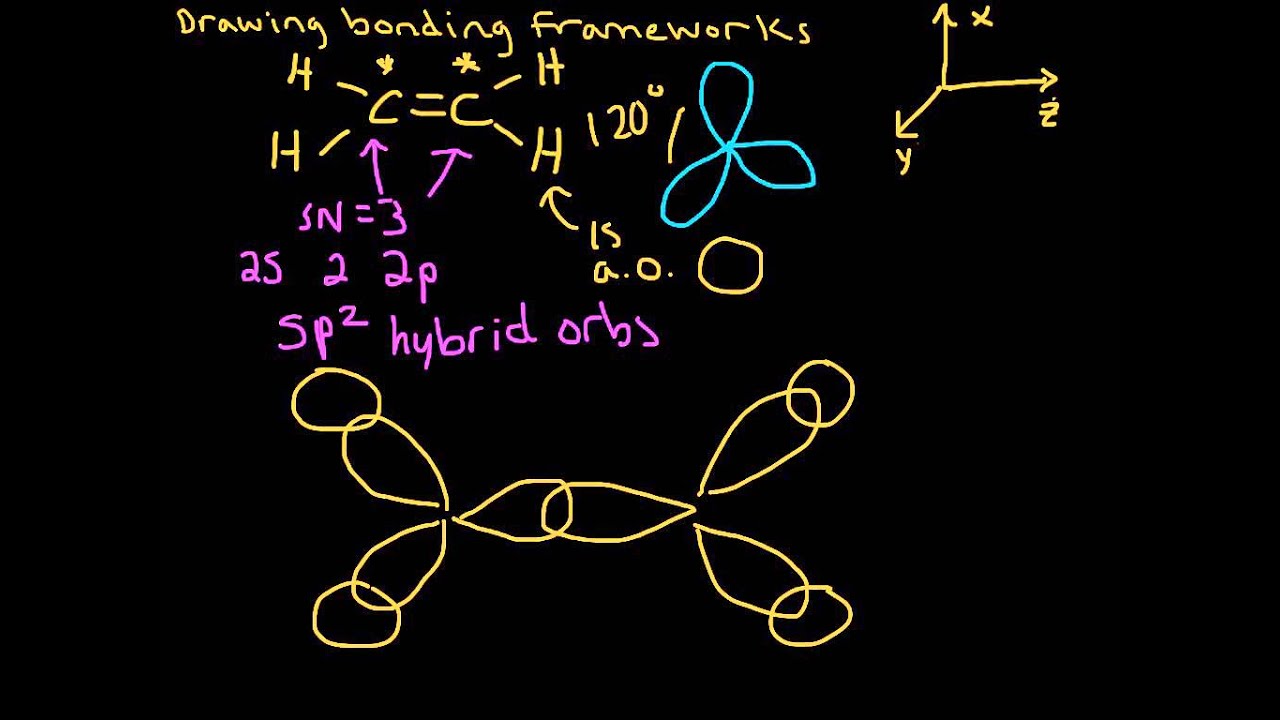
· what is the difference between the sentences below? Label the structure of c2h4, including overlapping orbitals, and label all bonds. Consider the representation of dashed lines in the orbital structure of ethene, c2h4, as they typically signify a type of bond formation involving unhybridized p-orbitals. The point group of ethene, c2h4, is d 2h. · a quick explanation of the molecular geometry of c2h4 including a description of the c2h4 bond angles. looking at the c2h4 lewis structure we can see that the. This reaction is ________. C2h4( g)+h2( g)→c2h6( g) δh =−137. 5 kj;δs =−120. 5 j/k calculate δg at 25∘c and determine whether the reaction is spontaneous. Labels can be used once, more than once, or not … A) spontaneous at all temperatures b) spontaneous only … · master the c2h4 lewis structure with our step-by-step guide and detailed diagram. Consider the equilibrium c2h6 (g) ⇌ c2h4 (g) + h2 (g). At 1000 k and a constant total pressure of 1 bar, c2h6 (g) is introduced into a reaction vessel. Welcome to geometry of molecules, and today in this video we are going to help you know the step-by-step method for determining the lewis structure of the c2h4 molecule. Mary decided to get pregnant before it was too late. Draw the lewis structures of c2h6, c2h4, and c2h2. draw the molecules by placing atoms on the grid and connecting them with bonds. The coordinate axes and the molecular structure are shown in figure 3. The enthalpy of formation of ethene, c2h4, at 1 atm, 25\deg c is 52,280 kj/kmol. Mary decided to get pregnant before it would be too late. Be sure to include all resonance structures that satisfy the octet rule. Learn how to draw the ethylene molecule, understand its electron geometry, and explore its … For the reaction c2h6 (g) → c2h4 (g) + h2 (g) δh° is +137 kj/mol and δs° is +120 j/k ∙ mol. (methene = 28. 05 kg/kmol). Express the free … Consider the following reaction: Vsepr names and atoms molecule or lon ccl4 # of valence electrons # of atoms or sets of lone pairs surrounding central atom structure geometry (name) 32 8 16 tetrahedral … Include all hydrogen atoms. · this can help us determine the molecular geometry, how the molecule might react with other molecules, and some of the physical properties of the molecule (like boiling point and surface tension). Draw the lewis structure for the ethylene (c2h4 molecule. Drag the appropriate labels to their respective targets. Assume all bond angles are 120 degree. determine the higher and lower heating values.
C2H4 Molecular Geometry: The Properties You Must Understand (Before It'S Too Late!)
· what is the difference between the sentences below? Label the structure of c2h4, including overlapping orbitals, and label all bonds. Consider the representation of...