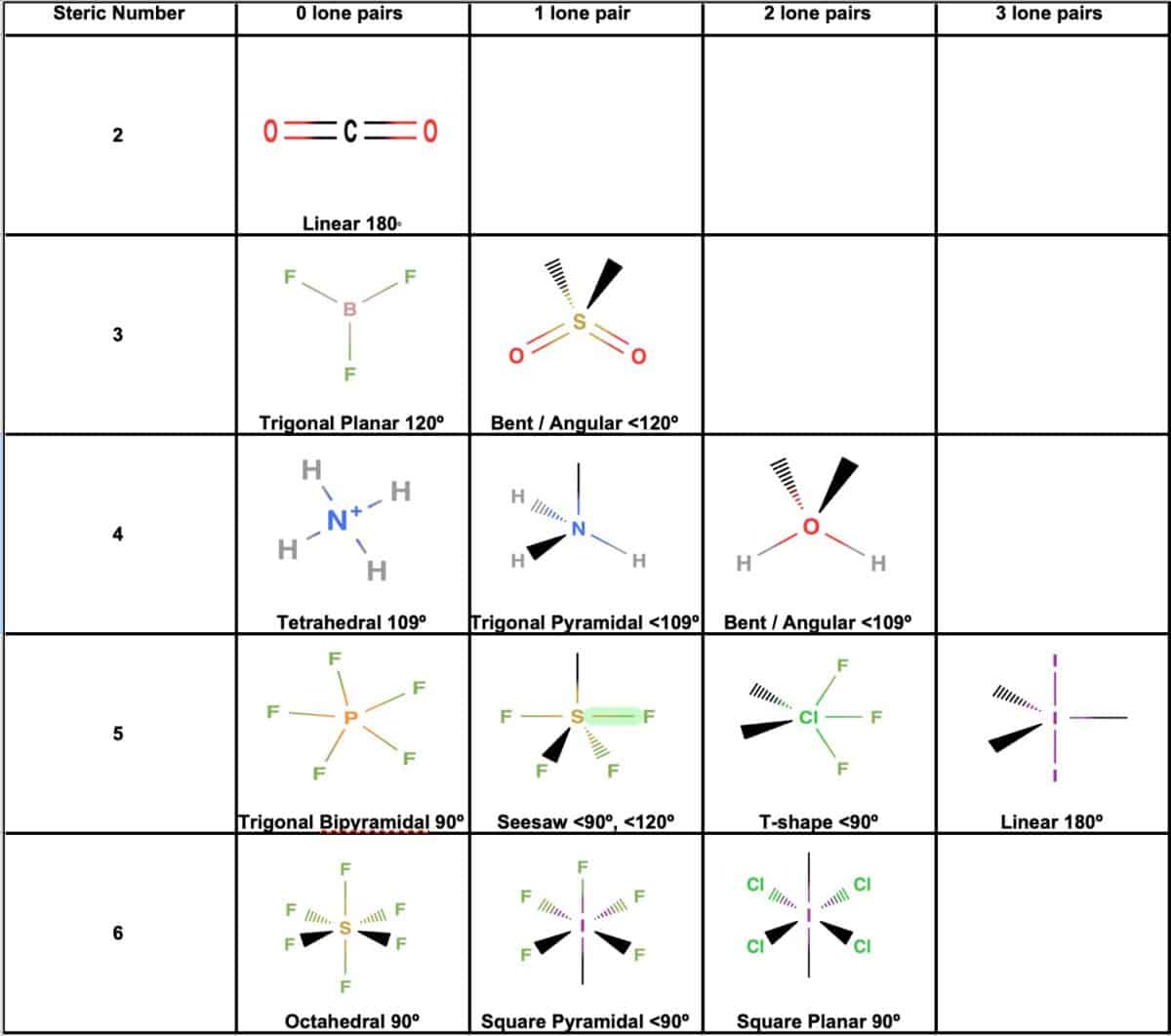
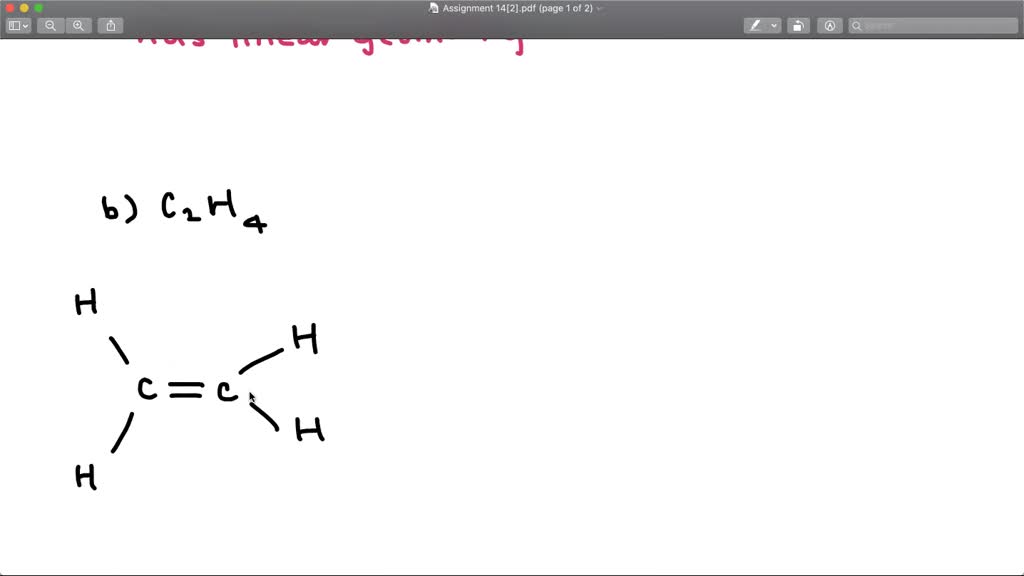
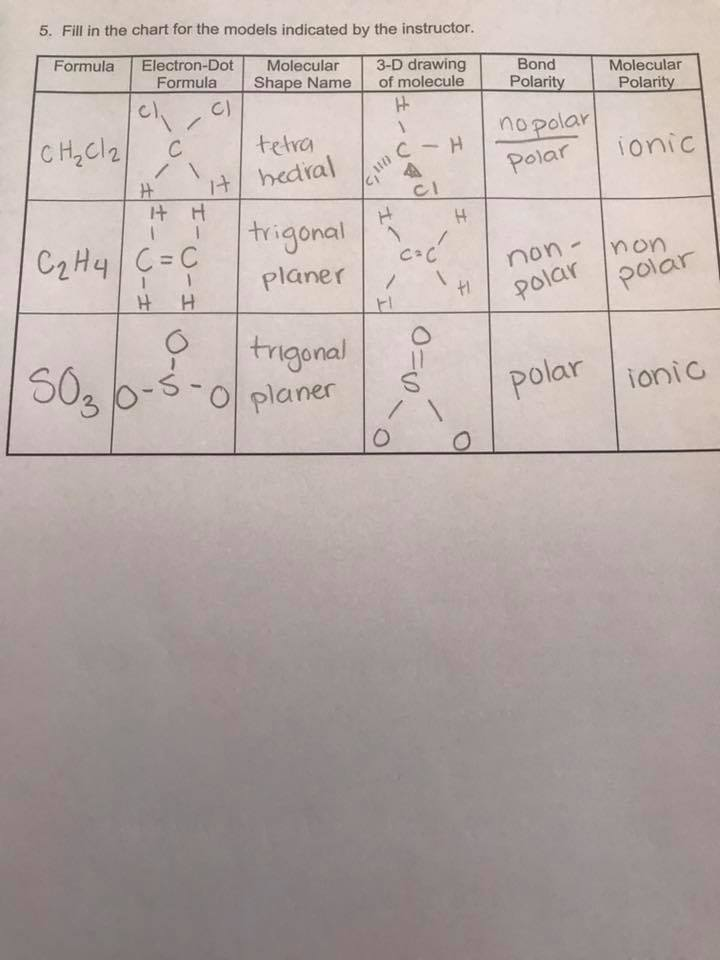
The enthalpy of formation of ethene, c2h4, at 1 atm, 25\deg c is 52,280 kj/kmol. Drag the appropriate labels to their respective targets. Draw the lewis structure for the ethylene (c2h4 molecule. determine the higher and lower heating values. Be sure to include all resonance structures that satisfy the octet rule. Draw the lewis structures of c2h6, c2h4, and c2h2. draw the molecules by placing atoms on the grid and connecting them with … Label the structure of c2h4, including overlapping orbitals, and label all bonds. The point group of ethene, c2h4, is d 2h. The coordinate axes and the molecular structure are shown in figure 3.
C2H4 Molecular Geometry: The Properties That Determine How It Behaves!
The enthalpy of formation of ethene, c2h4, at 1 atm, 25\deg c is 52,280 kj/kmol. Drag the appropriate labels to their respective targets. Draw the...