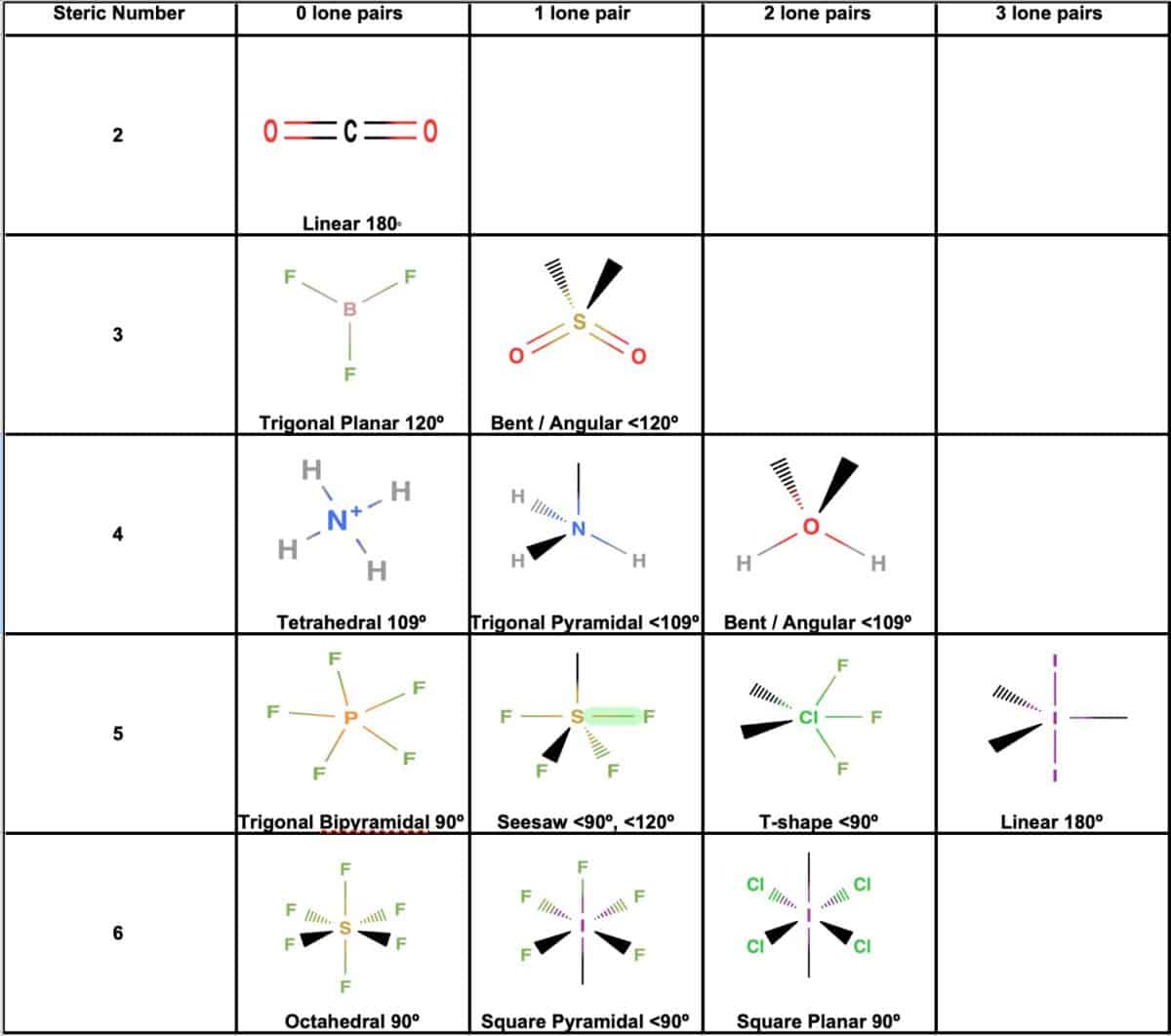
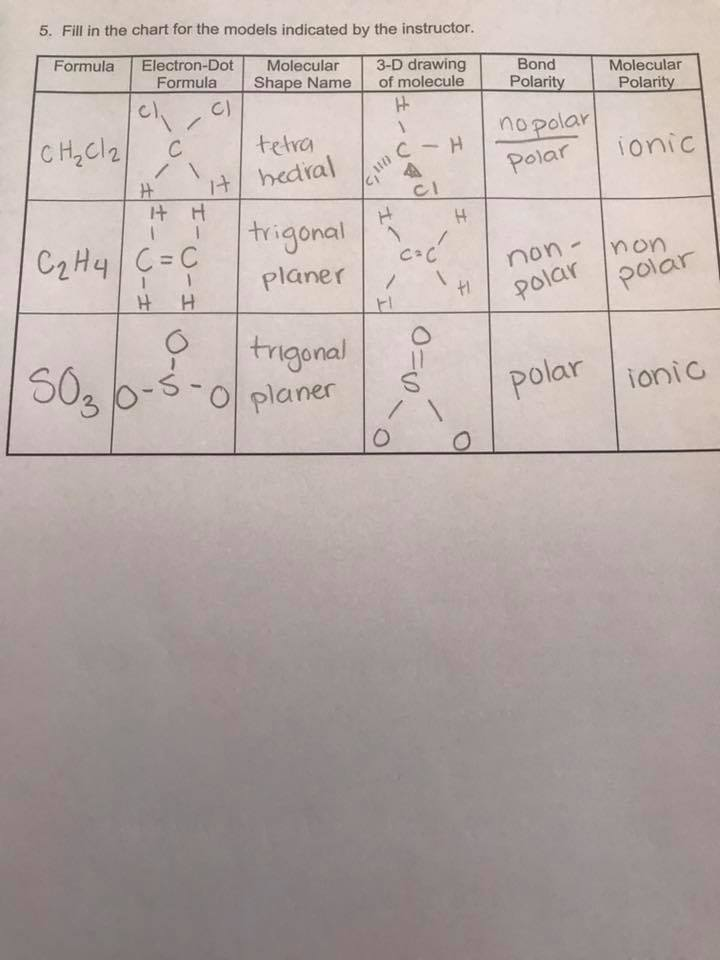
The point group of ethene, c2h4, is d 2h. determine the higher and lower heating values. Label the structure of c2h4, including overlapping orbitals, and label all bonds. A) spontaneous at all temperatures b) spontaneous only at high … Consider the representation of dashed lines in the orbital structure of ethene, c2h4, as they typically signify a type of bond formation involving unhybridized p-orbitals. Drag the appropriate labels to their respective targets. At 1000 k and a constant total pressure of 1 bar, c2h6 (g) is introduced into a reaction vessel. Draw the lewis structure for the ethylene (c2h4 molecule. Vsepr names and atoms molecule or lon ccl4 # of valence electrons # of atoms or sets of lone pairs surrounding central atom structure geometry (name) 32 8 16 tetrahedral h20 … We intend to construct … Include all hydrogen atoms. The enthalpy of formation of ethene, c2h4, at 1 atm, 25\deg c is 52,280 kj/kmol. The coordinate axes and the molecular structure are shown in figure 3. For the reaction c2h6 (g) → c2h4 (g) + h2 (g) δh° is +137 kj/mol and δs° is +120 j/k ∙ mol. Consider the equilibrium c2h6 (g) ⇌ c2h4 (g) + h2 (g). Draw the lewis structures of c2h6, c2h4, and c2h2. draw the molecules by placing atoms on the grid and connecting them with bonds. (methene = 28. 05 kg/kmol). Labels can be used once, more than once, or not at … C2h4( g)+h2( g)→c2h6( g) δh =−137. 5 kj;δs =−120. 5 j/k calculate δg at 25∘c and determine whether the reaction is spontaneous. Assume all bond angles are 120 degree. This reaction is ________. Express the free … Be sure to include all resonance structures that satisfy the octet rule. Consider the following reaction:
C2H4 Molecular Geometry: How Its Molecular Geometry Affects Its Properties
The point group of ethene, c2h4, is d 2h. determine the higher and lower heating values. Label the structure of c2h4, including overlapping orbitals,...