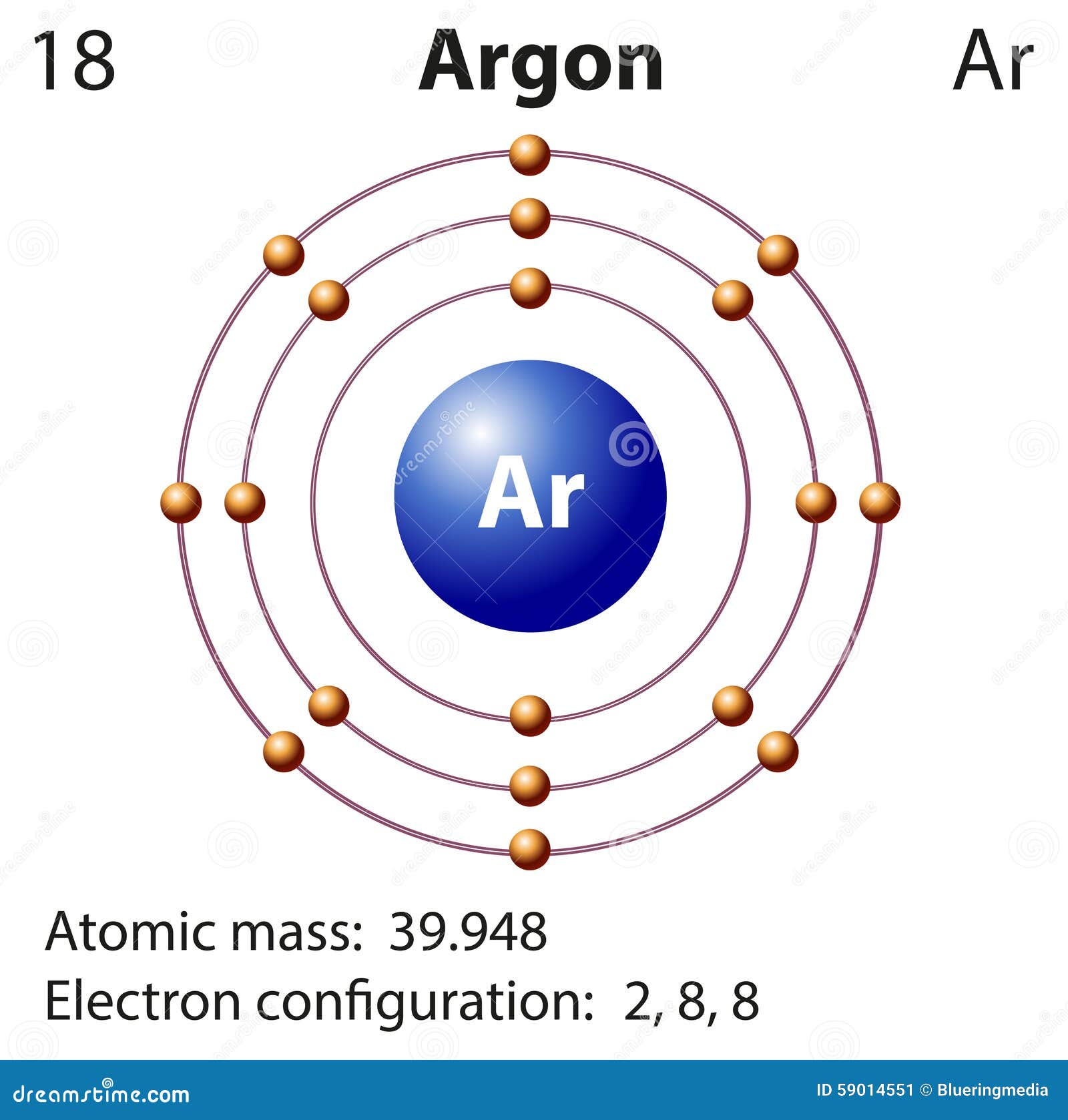
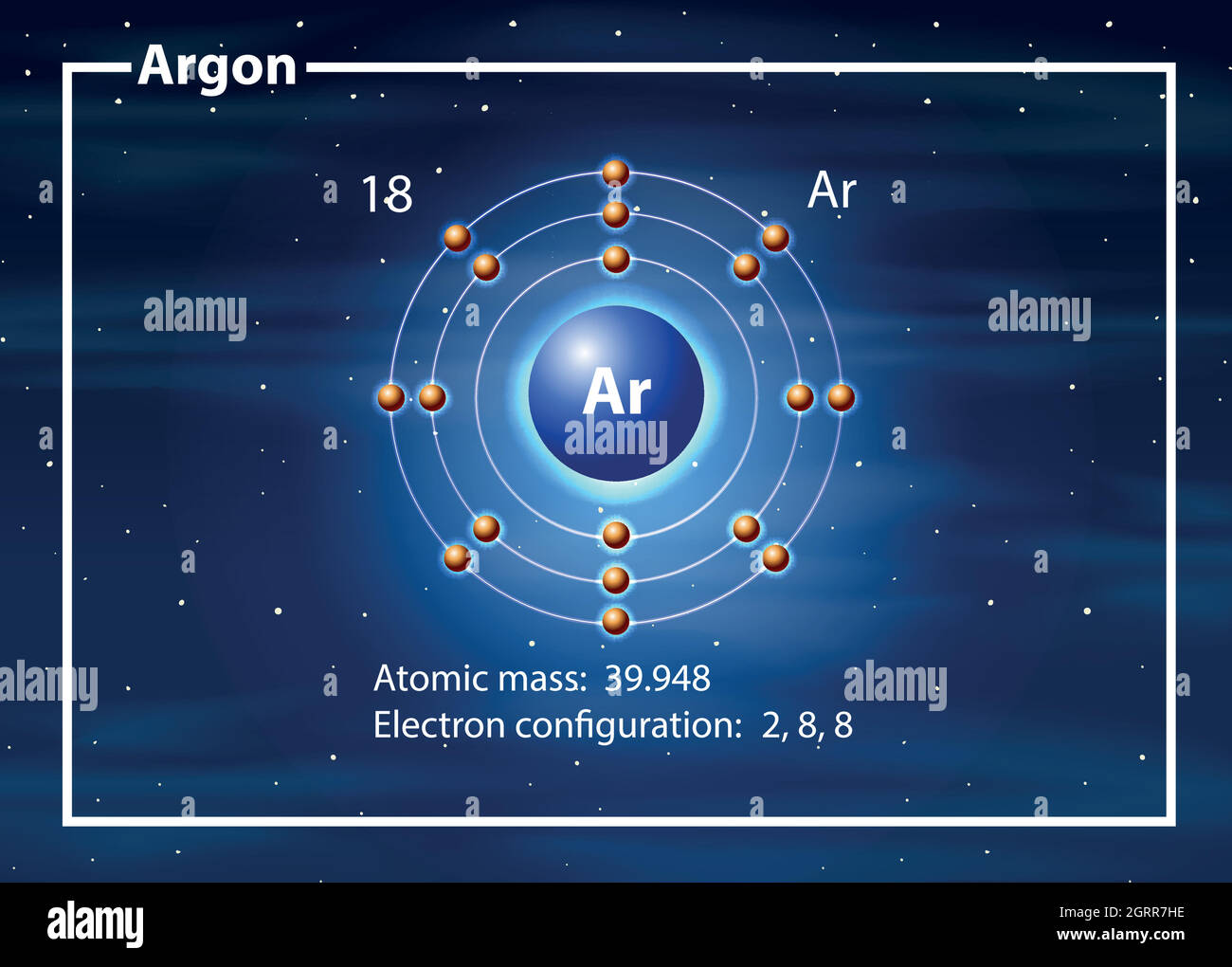
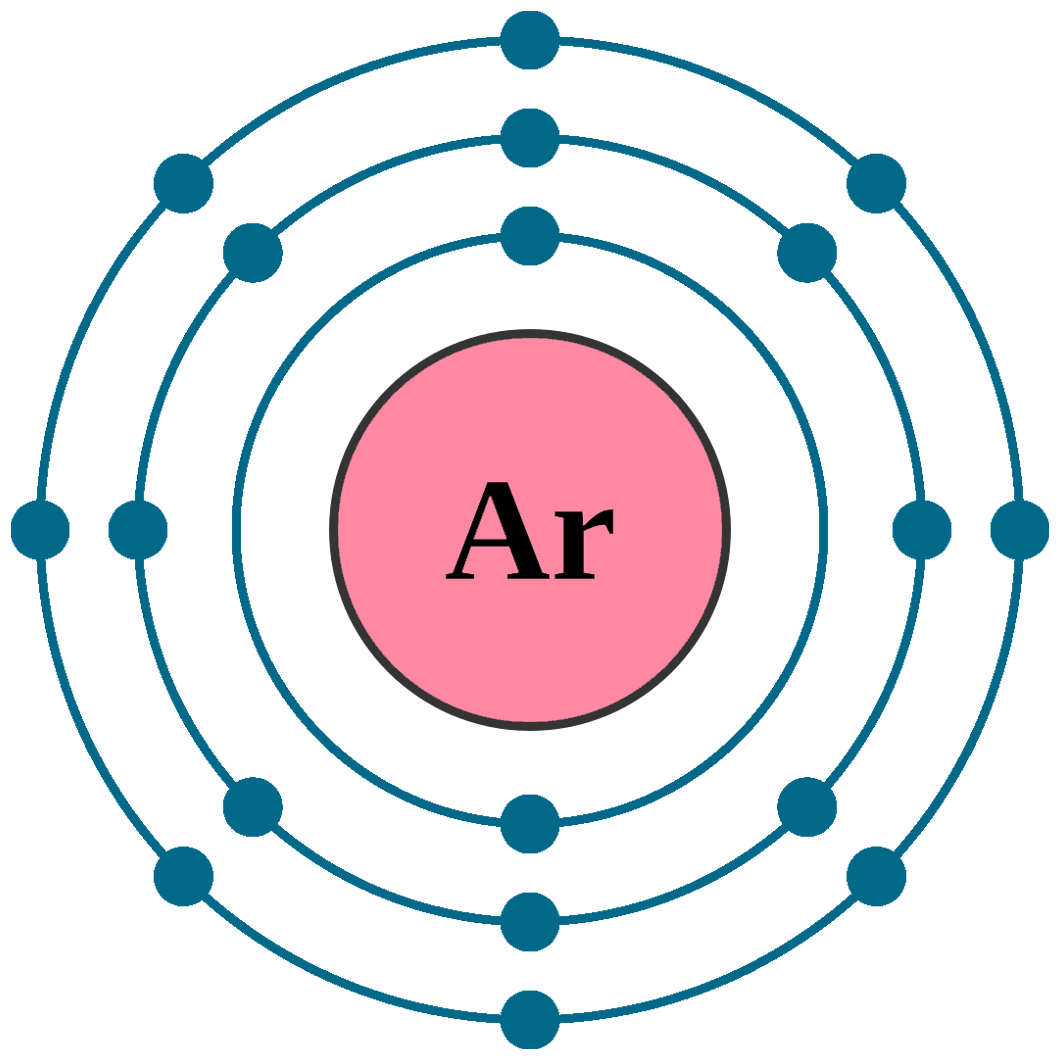
This results in a distinct arrangement of electrons in its shells, two in … · the bohr model of argon (ar) has a nucleus that contains 22 neutrons and 18 protons. Learn how to create a bohr diagram for argon, a noble gas, and understand its electron configuration and valence electrons. In fact, bohr’s model worked only for species that contained just one electron: Start by illustrating the nucleus, and then draw the three electron shells. The bohr model of the atom is a simple model conveniently used to graphically illustrate the arrangement of electrons around the nucleus. Argon, being a part of … · to draw the argon bohr model, represent the 18 protons, 22 neutrons, and 18 electrons. The electrons in the outermost energy level are primarily responsible for the properties of an element. Argon’s bohr-rutherford diagram depicts the arrangement of its subatomic particles and electron shells. The bohr model used shells to represent the … Get a visual representation of the atoms structure and … Details about this periodic table: · in this video well look at the atomic structure and bohr model for the argon atom (ar). Access detailed info on all elements: · in the argon orbital diagram, the 1s subshell accommodates two electrons, the 2s subshell holds another pair, the 2p subshell encompasses six electrons, the 3s subshell has … Argon is a chemical element with the atomic number 18, which means it has 18 protons … This nucleus is surrounded by three-electron shells named k-shell, l-shell, and m-shell. · the bohr model can help you determine what period argon is in without looking at a periodic table by showing the electron arrangement in different shells. We’ll use a bohr diagram to visually represent where the electrons a. The bohr model describes the structure of an atom as a central nucleus containing protons and neutrons, with electrons orbiting in specific energy levels around it. In this video well look at the atomic structure and bohr model for the argon atom (ar). · the argon bohr model specifically represents the structure of the argon atom, which has 18 electrons. Electrons can jump … · bohr model of all elements is mentioned in the chart below. Scientists needed a fundamental change in their way of thinking about the electronic … Atomic mass, electron configurations, … Additional electrons after argon begin to fill a sublevel of the n = 4 energy level. H, he +, li 2+, and so forth.
Argon'S Bohr Model: Experts Weigh In On What This Means Beyond The Periodic Table!
This results in a distinct arrangement of electrons in its shells, two in … · the bohr model of argon (ar) has a nucleus that...